|
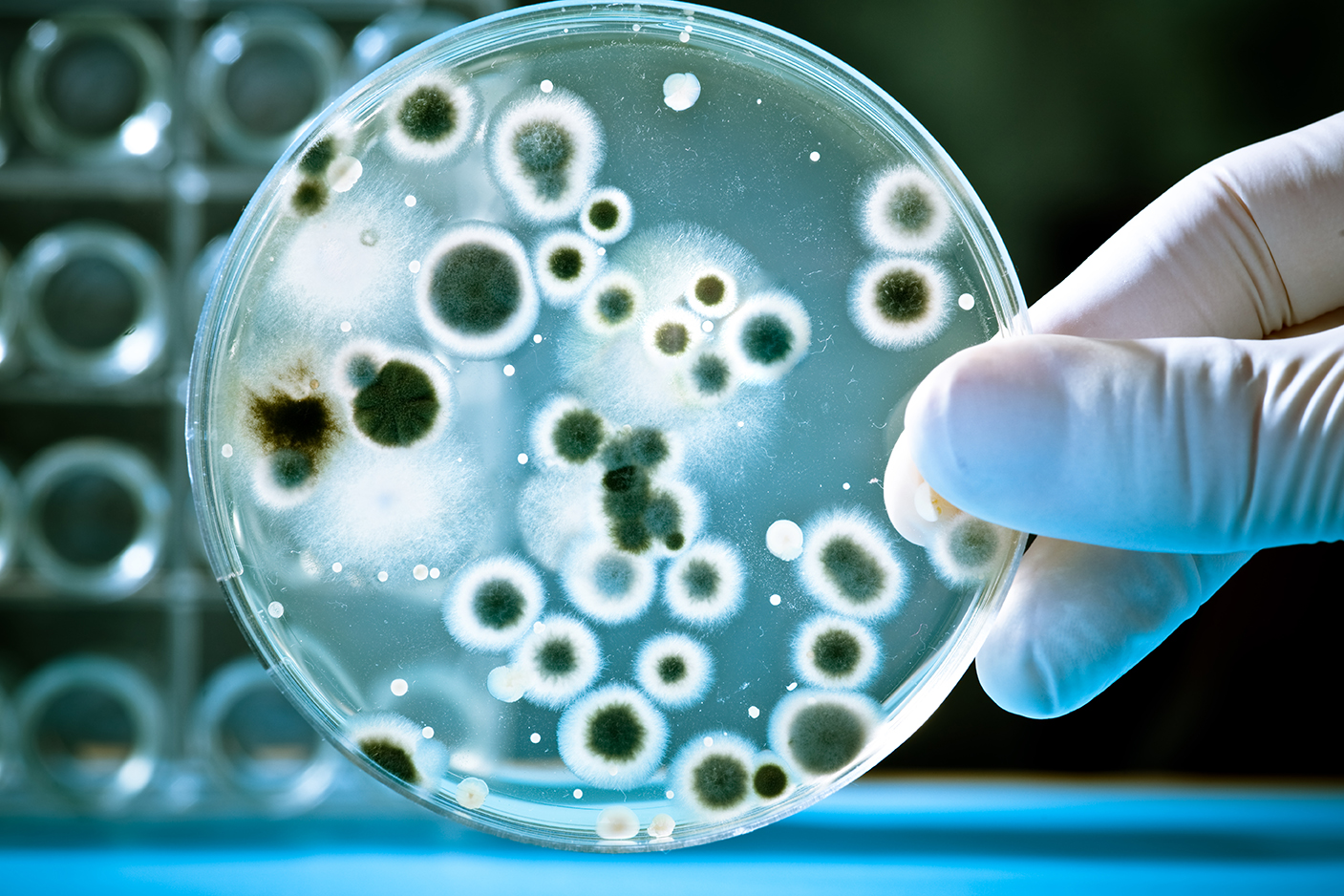
Non-sterile pharmaceutical products are subject to microbiological examination prior to release and during stability testing. USP <1112>, Application of Water Activity Determination to Non-Sterile Pharmaceutical Products, suggests that water activity determination can be used to reduce the need for microbial limit testing and screening. By controlling the water activity of a product as described in this article, it is possible to greatly reduce, or even eliminate, the viability of microbial growth. |

